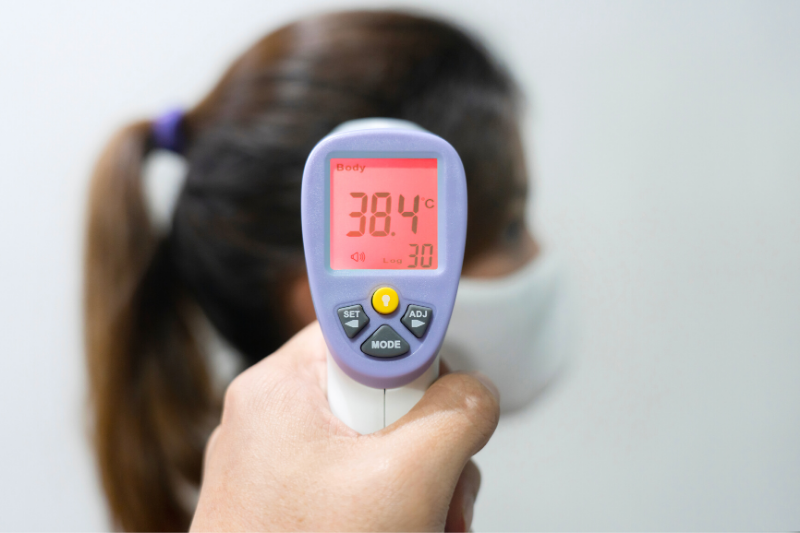
In a recent case handled by our firm, Customs officials insisted that the importer classify their product under the category that defines products for medical use, which requires the imported product to be submitted to ANVISA (Brazilian Health Regulatory Agency) for analysis in order to obtain an Import License (LI). This requirement was maintained even after ANVISA itself informed, through a consultation, that the thermometers in question are not subject to its approval.
“"Equipment used to measure the body temperature of people for the sole purpose of screening people in public environments, without indication for medical diagnostic purposes, is not considered a health product under RDC No. 185/2001, therefore it does not require authorization from ANVISA for importation purposes.".
Upon analyzing the matter, the Magistrate decided as follows in the context of urgent relief: “…I PARTIALLY GRANT THE PRELIMINARY INJUNCTION REQUEST in order to order the respondent Authority, or whoever acts in its place, to continue, within 5 days, the customs clearance procedures for the goods that are the subject of the Import Declarations, removing (i) the classification of the goods as health products and the requirements arising therefrom, as well as (ii) any legal consequences, releasing them thereafter, if these are the only obstacles to doing so.”
This is an important decision for companies that are importing infrared thermometers!
For more information, contact our team!
Decision commented by Michel Pereira, Lawyer. Areas of Expertise: Customs Law, Tax Law, and Corporate Law. OAB (Brazilian Bar Association): 295.435.



